
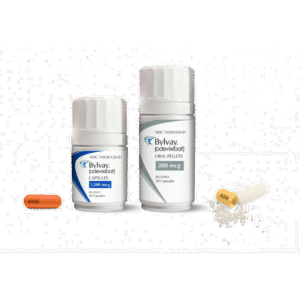
Buy Bylvay (odevixibat) Online
From $5,167.80
- DISEASE INDICATIONS: Progressive Familial Intrahepatic Cholestasis
- MANUFACTURER: Albireo Pharma
- USAGE: Oral
- MEDICINE APPROVED BY:
- European Medical Agency (EMA)
- Food and Drug Administration (FDA)
Bylvay, with the generic name odevixibat, is a prescription medication used in the treatment of certain rare and chronic liver diseases that involve impaired bile flow. Specifically, it is indicated for the treatment of pruritus (itching) associated with progressive familial intrahepatic cholestasis (PFIC) in patients as young as 6 months of age and older.

Buy Bylvay (odevixibat) Online
From $5,167.80
- Description
- Additional information
- Reviews (0)
Description
Bylvay, with the generic name odevixibat, is a prescription medication used in the treatment of certain rare and chronic liver diseases that involve impaired bile flow. Specifically, it is indicated for the treatment of pruritus (itching) associated with progressive familial intrahepatic cholestasis (PFIC) in patients as young as 6 months of age and older. PFIC is a group of genetic disorders that affect bile ducts in the liver, leading to impaired bile flow and a range of symptoms, including severe itching. Bylvay helps regulate bile flow, reducing the severity of pruritus and improving quality of life. Here is a detailed description of Bylvay:
Drug Class: Bylvay is classified as a bile acid transporter inhibitor. It targets specific bile acid transporters in the liver, modulating bile flow and reducing the accumulation of toxic bile acids.
Indications: Bylvay is indicated for the treatment of pruritus (itching) associated with progressive familial intrahepatic cholestasis (PFIC) in patients aged 6 months and older. PFIC is a rare and genetic liver disorder characterized by impaired bile flow due to mutations in specific genes.
Mechanism of Action: Odevixibat, the active ingredient in Bylvay, works by inhibiting the ileal bile acid transporter (IBAT) in the intestines. By doing so, it reduces the reabsorption of bile acids, leading to an increase in the secretion of bile acids into the intestines. This action helps decrease the accumulation of toxic bile acids in the liver, which is a characteristic feature of PFIC.
Dosage and Administration: The dosage of Bylvay is determined by a healthcare provider based on the individual patient’s age, weight, and specific clinical needs. It is administered as an oral suspension and should be taken once daily with or without food.
Effectiveness: Clinical trials have shown that Bylvay can effectively reduce pruritus and improve quality of life in patients with PFIC by regulating bile flow and reducing the accumulation of toxic bile acids.
Side Effects: Like all medications, Bylvay may have side effects. Common side effects may include diarrhea, abdominal pain, and vomiting. Patients receiving Bylvay should be monitored for potential side effects and adverse reactions.
Contraindications: Bylvay is contraindicated in individuals with known hypersensitivity to odevixibat or any of its components.
Consultation with a Healthcare Provider: Before starting Bylvay, individuals should consult with their healthcare provider to determine if it is a suitable treatment option and to discuss potential benefits and risks. Regular monitoring and follow-up are essential during treatment with Bylvay to assess treatment effectiveness and manage potential side effects.
Bylvay represents an important therapeutic option for individuals with progressive familial intrahepatic cholestasis (PFIC) who experience severe pruritus as a result of impaired bile flow. Its use should always be under the guidance and supervision of qualified healthcare professionals who can assess the individual’s specific medical condition and needs.
Additional information
| Package | 30 hard capsule of 200 μg, 30 hard capsule of 400 μg, 30 hard capsule of 1,200 μg |
|---|
Be the first to review “Buy Bylvay (odevixibat) Online” Cancel reply
Related Products

Buy Ocaliva (obeticholic acid) Online
Total Sales: 0
SKU: 384764



Reviews
There are no reviews yet.